|
Organic Versus Inorganic Compounds. There are chief differences between organic and inorganic compounds. While both types of compounds make up the basis of chemistry, the two types are rather different. The main difference is in the presence of a carbon atom; organic compounds will contain a carbon atom (and often a hydrogen atom, to form hydrocarbons), while almost all inorganic compounds do not contain either of those two atoms. While most inorganic compounds do not contain carbon, there are a few that do. Carbon monoxide and carbon dioxide, for example, each contain carbon atoms, but the amount is not large enough to form strong bonds with the oxygen present in the molecule. Due to the small amount of carbon and the weak bonds it forms, scientists have long classified those molecules as inorganic, but this has led some in the scientific community to declare the need for a better classification system for compounds. Another important distinction between organic and inorganic compounds is the type of molecule and its association with living things. Organic compounds will include things like the nucleic acids, found in DNA, lipids and fatty acids found in the cells of living organisms, proteins and enzymes that are necessary for cellular processes to take place, and more. Meanwhile, inorganic compounds include the salts, metals, and other elemental compounds. C1. 2H2. 2O1. 1 - Sucrose, better known as the sugar we use at home, is an important organic compound that contains not only carbon and hydrogen, but contains them in abundance and in ratios greater than the oxygen present. CH4 - Also known by the more common name methane, this carbon and hydrogen based organic compound is a widely recognized waste product of living things. C5. 5H7. 2O5. N4. Mg - Better known among researchers as chlorophyll- a, this component found in green plants not only is high in carbon and hydrogen ratios, but is also a chief factor in the process of photosynthesis in plants. 
Ca. Cl. 2 - Calcium chloride is an inorganic compound with a variety of uses, but its lack of either a carbon atom, a hydrogen atom, or both means it is classified as an inorganic compound. What are some examples of common household organic compounds? Vinegar (acetic acid, CH3. COOH) * Baking soda - sodium bicarbonate H2. NO3 (base) Cooking spray - its a hydrocarbon, usually ethane CH3. CH3 * The gas you cook with is an organic compound (hydrocarbon). Volatile organic compounds change easily from liquid form to vapor. What are volatile organic compounds (VOCs)? Organic compounds are chemicals that.   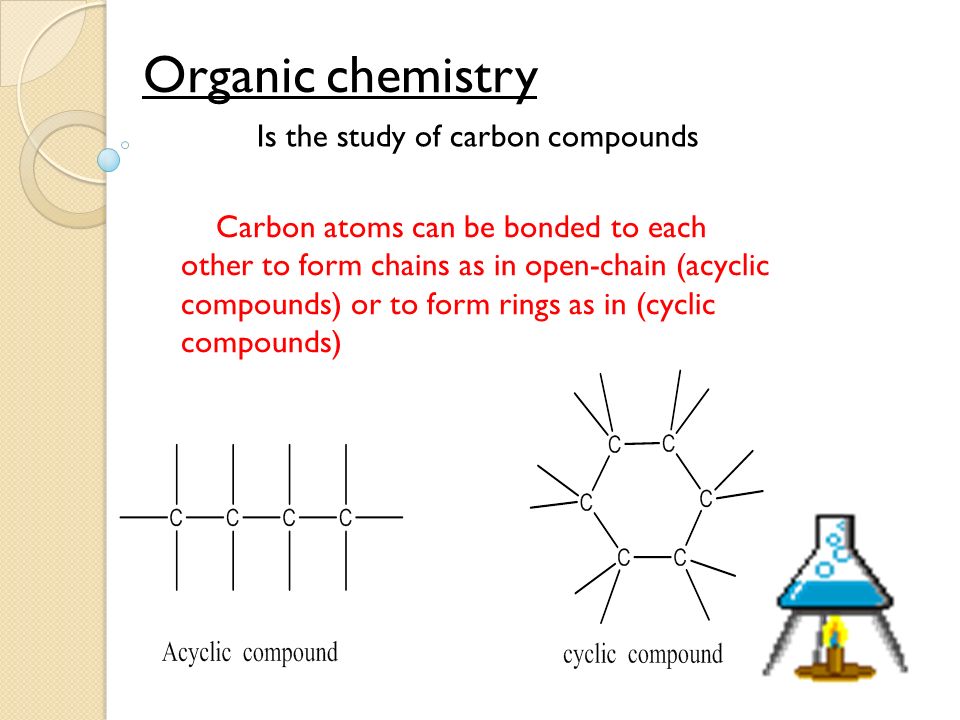 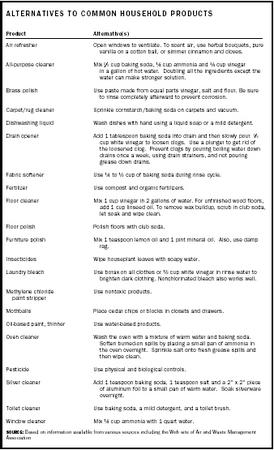 
Inorganic Chemicals and Compounds Information. Bio-Inorganic Compounds - natural and synthetic compounds that include metallic. Inorganic compound This article has multiple issues. Please help improve it or discuss these. C - Organic Compounds. Chemical Structures Starting with the Letter Q. Steroids - Molecular Structures. Chemical Structures Starting with the Letter L.  
What are example of covalent compounds that can be found in household items? In the organic compounds, the (H) atoms cannot be less than (C) atoms. Learn about household cleaning products from the Cleveland Clinic, including chemical ingredients found in the garage, kitchen, utility room, and more. Some products release dangerous chemicals, including volatile organic compounds. When using cleaning or household products, keep the area well ventilated. Examples of organic compounds found in households.there are lots of things found inside the house but what things are organic compounds?
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
August 2017
Categories |

 RSS Feed
RSS Feed
